Aldehyde Ketone And Carboxylic Acid MCQ Chapter 12 Question 1.
Which of the following compounds will give butanone on oxidation with alkaline KMnO4 solution ?
(A) Butan-l-ol
(B) Butan-2-ol
(C) Both of these
(D) None of these
Answer:
(B) Butan-2-ol
Explanation:
Butan-2-ol is secondary alcohol which on oxidation with alkaline KMnO4 solution gives butanone (ketone).
Aldehydes And Ketones Class 12 MCQ Chapter 12 Question 2.
Write the IUPAC name of
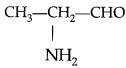
(A) 1-Aminopropanaldehyde
(B) 2-Aminopropanal
(C) 1-Aminoethan-l-al
(D) None of the above
Answer:
(B) 2-Aminopropanal
Explanation:
2-Aminopropanal
MCQ On Aldehydes Ketones And Carboxylic Acids Chapter 12 Question 3.
What kind of compounds undergo Cannizaro reactions ?
(A) Ketones with no a- hydrogen
(B) Aldehydes with a- hydrogen
(C) Carboxylic acids with a- hydrogen
(D) Aldehydes with no a- hydrogen
Answer:
(D) Aldehydes with no a- hydrogen
Explanation:
Aldehydes with no a-hydrogen undergo Canizzaro reaction.
Class 12 Chemistry Chapter 12 MCQ Chapter 12 Question 4.
Write the product(s) in the following reactions:
(A) No product formed
Answer:
Option (B) is correct.
Explanation:
It is a nucleophilic addition reaction.
Aldehydes, Ketones And Carboxylic Acids MCQ With Answers Pdf Question 5.
Compounds A and C in the following reaction are
(A) identical
(B) positional isomers
(C) functional isomers
(D) optical isomers
Answer:
(B) positional isomers
Explanation:
In compound A and C, position of -OH group is changed. So, these are positional isomers.
MCQ Of Aldehydes And Ketones Class 12 Chapter 12 Question 6.
In Clemmensen reduction carbonyl compound is treated with ………….
(A) zinc amalgam + HCl
(B) sodium amalgam + HCl
(C) zinc amalgam + nitric acid
(D) sodium amalgam + HNO3
Answer:
(A) zinc amalgam + HCl
Explanation:
Clemmensen reduction is used to convert carbonyl group to CH2 group as follows:
MCQ Of Aldehyde And Ketone Class 12 Chapter 12 Question 7.
The reagent which does not react with both, acetaldehyde and benzaldehyde.
(A) Sodium hydrogen sulphite
(B) Phenyl hydrazine
(C) Fehling’s solution
(D) Grignard reagent
Answer:
(C) Fehling’s solution
Explanation:
Aliphatic aldehydes(acetaldehyde) reduce the Fehling’s solution to red cuprous oxide.
Aromatic aldehydes (benzaldehyde) do not react with Fehling’s solution.
Aldehydes Ketones And Carboxylic Acids MCQ Question 8.
![]()
(A) C6H5COOH + CH4
(B) C6H5COONa + CHI3
(C) C6H6 + CH3COONa + HI
(D) C6H5CH2COOH
Answer:
(B) C6H5COONa + CHI3
Explanation:
MCQs On Aldehydes And Ketones Class 12 Chapter 12 Question 9.
Predict the product of the following reaction:
(A)CH3CH2CH3
(B) CH3CHOHCH3
(C) CH3CH2CHO
(D) CH3CONHCH3
Answer:
(A)CH3CH2CH3
Explanation:
It is a VVolff-Kishner reduction which converts ![]() group into-CH2– group.
group into-CH2– group.
Aldehydes And Ketones MCQ Class 12 Chapter 12 Question 10.
Which of the following compounds is most reactive towards nucleophilic addition reactions ?
Answer:
Option (A) is correct.
Explanation:
Methyl benzaldehyde < Benzal-dehyde < Propanone < Ethanal – reactivity to-wards nucleophilic substitution. Aldehydes are more reactive than aliphatic ketones. Aliphatic ketones are more reactive than aromatic ketones. The +1 effect is more in ketone than in aldehyde. Thus ketone will be least reactive in nucleophilic addition reactions. The presence of electron withdrawing group increases the reactivity towards the addition while the presence of electron donating group decreases the reactivity of compound towards nucleophilic addition. Benzaldehyde does not favour nucleophilic addition reaction due to resonance stabilisation.
MCQ Aldehydes And Ketones Class 12 Chapter 12 Question 11.
Formaldehyde reacts with methyl magnesium bromide followed by hydrolysis to form.
(A) Methanol
(B) Ethanol
(C) Propanol
(D) Butanol
Answer:
(B) Ethanol
Explanation:
Aldehyde And Ketone MCQ Class 12 Chapter 12 Question 12.
Common name of Ethane-1,2-dioic acid is known as
(A) Oxalic acid
(B) Phthalic acid
(C) Adipic acid
(D) Acetic acid
Answer:
(A) Oxalic acid
Explanation:
Structural formula of Ethane-1, 2 dioicacidis
It is oxalic acid.
Aldehyde Ketone And Carboxylic Acid MCQ Questions Question 13.
The carboxylic acid that does not undergo HVZ reaction is
(A) CH3COOH
(B) (CH3)2COOH
(C) CH3CH2CH2CH2COOH
(D) (CH3)3CCOOH
Answer:
(C) CH3CH2CH2CH2COOH
Explanation:
The carboxylic acids having a-hydrogen atom undergo HVZ reaction. Since (CH3)3C. COOH doesnot contain a-H-atom; so, it does not undergo HVZ reaction.
Multiple Choice Questions Aldehydes And Ketones Question 14.
Which of the following acids does not form anhydride ?
(A) Formic acid
(B) Acetic acid
(C) Propionic acid
(D) n-butyric acid
Answer:
(A) Formic acid
Explanation:
Formic acid(HCOOH) does not form anhydride because it does not contain u-C-atom.
Aldehydes And Ketones MCQs Class 12 Chapter 12 Question 15.
Which of the following is the strongest acid?
(A) Acetic acid
(B) Phenol
(C) Methyl alcohol
(D) Water
Answer:
(A) Acetic acid
Explanation:
Acetic acid is the strongest acid because it loses H+ ion to form carboxylic ion (CH3COO– ) which gets stabilised by resonance:
Aldehydes Ketones And Carboxylic Acids Class 12 MCQ Question 16.
The reaction in which the aqueous solution of sodium salt of carboxylic acids on electrolysis give alkanes:
(A) Soda lime decarboxylation
(B) Kolbe’s electrolysis decarboxylation
(C) Dry distillation of calcium formate
(D) Reduction of carboxylic acid.
Answer:
(B) Kolbe’s electrolysis decarboxylation
Explanation:
It is Kolbe’s electrolytic decarboxylation.
RCOONa(aq) → RCOO– + Na+
At anode, 2RCOO → R-R + 2CO2 + 2e– Alkane
At cathode, 2H2 O + 2e– + H2 + 2OH–
Aldehydes And Ketones Questions And Answers Pdf Class 12 Question 17.
Arrange the following compounds in increasing order of acid strength
(A) (i) > (ii) > (iii)
(B) (ii) < (i) < (iii)
(C) (iii) < (i) < (ii)
(D) (iii) > (i) > (ii)
Answer:
(C) (iii) < (i) < (ii)
Explanation:
The electron with drawing group (-NO2) increases the acid strength of aromatic acids while electron releasing group (-CH3) decreases the acid strength of aromatic acids. Hence, the increasing order of add strength is given as
Aldehydes And Ketones MCQ Pdf Class 12 Chapter 12 Question 18.
Complete the following reaction: CH3COONa + NaOH-
Answer:
Option (C) is correct.
Explanation:
Question 19.
Identify the name of the given reaction:
(A) Etard reaction
(B) Hell-Volhard-Zelinsky reaction
(C) Stephen reaction
(D) None of the above
Answer:
(B) Hell-Volhard-Zelinsky reaction
Explanation:
Hell-Volhard-Zelinsky
Assertion And Reason Based MCQs
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is NOT the correct explanation of A
(C) A is true but R is false
(D) A is false and R is true
Question 1.
Assertion (A): Oxidation of ketones is easier than aldehydes.
Reason (R): C-C bond of ketones is stronger than C-H bond of aldehydes.
Answer:
(D) A is false and R is true
Explanation:
Oxidation of aldehydes are easier than ketones.
Question 2.
Assertion (A): Benzaldehyde is less reactive than ethanal towards nucleophilic addition reactions.
Reason (R): Ethanal is more sterically hindered.
Answer:
(B) Both A and R are true but R is NOT the correct explanation of A
Explanation:
The carbon atom of the carbonyl group of benzaldehyde is less electrophilic than carbon atom of carbonyl group present in elhanal. The polarity of the carbonyl group is reduced in benzaldehyde due to resonance hence it is less reactive than ethanal towards nucleophilic addition reaction.
Question 3.
Assertion (A): Aromatic aldehydes and formaldehyde undergo Cannizzaro reaction.
Reason (R): Aromatic aldehydes are almost as reac¬tive as formaldehyde.
Answer:
(C) A is true but R is false
Explanation:
Aromatic aldehydes and formalde¬hyde do not contain a-hydrogen and thus undergo Cannizzaro reaction. Formaldehyde is more reactive than aromatic aldehydes.
Question 4.
Assertion (A): Aldehydes and ketones, both react with Tollen’s reagent to form silver mirror.
Reason (R): Both aldehydes and ketones contain a carbonyl group.
Answer:
(D) A is false and R is true
Explanation:
Both aldehydes and ketones have carbonyl group but only aldehydes react with Tollens’ reagent to give silver mirror.
Question 5.
Assertion (A): Benzoic acid does not undergo Friedel-craft’s reaction.
Reaction (R): The carboxyl group is activating and undergo electrophilic substitution reaction.
Answer:
(C) A is true but R is false
Explanation:
The carboxyl group (-COOH) is deactivating group because it is electron with drawing group. It decreases the electron density at benzene ring, hence deactivates it towards electrophilic substitution reactions.
Question 6.
Assertion (A): Compounds containing -CHO group are easily oxidised to corresponding carboxylic acids.
Reason (R): Carboxylic acids can be reduced to alcohols by treatment with LiAlH4.
Answer:
(B) Both A and R are true but R is NOT the correct explanation of A
Explanation:
Compounds containing -CHO group are easily oxidised to corresponding* carboxylic acids.
Question 7.
Assertion (A): Aromatic carboxylic groups do not undergo Friedel- Crafts reaction.
Reason (R): Carboxyl group is deactivating and the catalyst aluminium chloride gets bonded to the carboxyl group.
Answer:
(A) Both A and R are true and R is the correct explanation of A
Explanation:
Aromatic carboxylic groups do not urldergo Friedel-Crafts reaction because Carboxyl group is deactivating and the catalyst aluminium chloride gets bonded to the carboxyl group.
Question 8.
Assertion (A): Carboxylic acids are more acidic than phenols.
Reason (R): Phenols are ortho and para directing.
Answer:
(B) Both A and R are true but R is NOT the correct explanation of A
Explanation:
Carboxylic acids are more acidic than phenols as the carboxylate ion, the conjugate base of carboxylic acid is stabilized by two equivalent structures. Thus, the negative charge is delocalized effectively. However, in phenols, negative charge is less effectively delocalized over oxygen atom and carbon atoms in phenoxide ion.
Case-Based MCQs
I. Read the passage given below and answer the following questions:
Reduction of carboxylic acids and their derivatives plays an important role in organic synthesis, in both laboratory and industrial processes. Traditionally, the reduction is performed using stochiometric amounts of hydride reagents, generating stochiometric amounts of waste. A much more attractive, atom-economical approach is a catalytic reaction using H2; however, hydrogenation of carboxylic acid derivatives under mild conditions is a very challenging task, with amides presenting
the highest challenge among all classes of carbonyl compounds. Very few examples of the important hydrogenation of amides to amines, in which the C-O bond is cleaved with the liberation of water (Scheme 1), were reported. C-O cleavage of amides can also be affected with silanes as reducing agents. (Generation of amides to the with cleavage of the C-N products of C-O cleavage the case of anilides). The and neutral, homogeneous
Scheme 1. General Sche C-O cleavage
We have now prepared the new, dearomatized, bipyridine-based pincer complex 3, catalyst 3 (Here refered as Cat. 3). Remarkably, it efficiently catalyzes the selective hydrogenation of amides to form amines and alcohols (eq 1). The reaction proceeds under mild pressure and neutral conditions, with no additives being required.
Since the reaction proceeds well under anhydrous conditions, hydrolytic cleavage of the amide is not involved in this process. been reported.6 Amines and chemical, pharmaceutical and ch a reaction is conceptually step in amide hydrogenation bonvl group to form a very anhydrous condition involved in this pro
In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
(A) Assertion and reason both are correct statements and reason is correct explanation for assertion.
(B) Assertion and reason both are correct statements but reason is not correct explanation for assertion.
(C) Assertion is correct statement but reason is wrong statement.
(D) Assertion is wrong statement but reason is correct statement.
Question 1.
Assertion (A): The use of catalyst 3 is an efficient method of preparation of primary amines
Reason (R): Use of catalyst 3 is a step down reaction.
Answer:
(B) Assertion and reason both are correct statements but reason is not correct explanation for assertion.
Question 2.
Assertion (A): Use of hydride catalyst or hydrogen brings about cleavage of C-O bond in amides.
Reason (R): Hydride catalyst or hydrogen cause to reduction of amides.
Answer:
(B) Assertion and reason both are correct statements but reason is not correct explanation for assertion.
Question 3.
Assertion (A): N-methyl ethanamide on reaction with catalyst 3 will yield ethanol and methanamine.
Reason (R): Use of Catalyst 3 brings about cleavage of C-N bond of amides
Answer:
(A) Assertion and reason both are correct statements and reason is correct explanation for assertion.
Question 4.
Assertion (A): Aniline can be prepared from suitable amide using catalyst 3
Reason (R): The use of catalyst 3 is limited to aliphatic amides only.
Answer:
(C) Assertion is correct statement but reason is wrong statement.
II. Read the passage given below and answer the following questions:
Aldehydes, ketones and carboxylic acids are few of the major classes of organic compounds containing carbonyl group. Aldehydes are prepared by dehydrogenation or controlled oxidation of primary alcohols and controlled or selective reduction of acyl halides. Ketones are prepared by oxidation of secondary alcohols and hydration of alkynes. Carboxylic acids are prepared by the oxidation of primary alcohols, aldehydes and alkenes by hydrolysis of nitriles and by treatment of Grignard reagents with carbon dioxide.
Question 1.
Name a method by which both aldehydes and ketones can be prepared.
(A) Reduction of carboxylic acids
(B) Ozonolysis of alkenes
(C) Oxidation of alcohols
(D) All of the above
Answer:
(D) All of the above
Explanation:
Both aldehydes and ketones can be prepared by all these methods.
Question 2.
How will you distinguish between aliphatic aldehydes and aromatic aldehydes ?
(A) Fehling’s test
(B) Benedict’s test
(C) Iodoform test
(D) Hinsberg reagent
Answer:
(A) Fehling’s test
Explanation:
On heating an aldehyde with Fehling’s reagent, a reddish brown precipitate is obtained. Aldehydes are oxidised to corresponding carboxvlate anion. Aromatic aldehydes do not respond to this test.
Question 3.
Name the main compounds A and B formed in the following reaction:
![]()
(A) CH3CH2COOH [A], CH3CH2CH3
(B) CH3CH2CHO [A], C2H4 [B]
(C) CH3COCH3 [A], CH3CH2CH3 [B]
(D) CH3COCH3 [A], C2H6 [B]
Answer:
(C) CH3COCH3 [A], CH3CH2CH3 [B]
Explanation:
Question 4.
The reagent which does not react with both, acetone and benzaldehyde.
(A) Sodium hydrogensulphite
(B) Phenyl hydrazine
(C) Fehlings’ solution
(D) Grignard reagent
Answer:
(C) Fehlings’ solution
Explanation:
Fehling’s solution does not react with acetone and benzaldehyde as aromatic I aldehydes and ketones do not react with Fehling’s solution. :
OR
Through which of the following reactions number of carbon atoms can be increased in the chain?
(A) Grignard reaction
(B) Cannizzaro reaction
(C) Clemmenson reduction
(D) HVZ reaction
Answer:
(A) Grignard reaction
Explanation:
The number of C-atoms can be i increased in the chain by Grignard reaction.
III. Read the passage given below and answer the following questions:
Reductive alkylation is the term applied to the process of introducing alkyl groups into ammonia or a primary or secondary amine by means of an aldehyde or ketone in the presence of a reducing agent. The present discussion is limited to those reductive alkylations in which the reducing agent is hydrogen and a catalyst or “nascent” hydrogen, usually from a metalacid combination; most of these reductive alkylations have been carried out with hydrogen and a catalyst.
The principal variation excluded is that in which the reducing agent is formic acid or one of its derivatives; this modification is known as the Leuckart reaction. The process of reductive alkylation of ammonia consists in the addition of ammonia to a carbonyl compound and reduction of the addition compound or its dehydration product. The reaction usually is carried out in ethanol solution when the reduction is to be effected catalytically:
Since the primary amine is formed in the presence of the aldehyde it may react in the same way as ammonia, yielding an addition compound, a Schiff’s base (RCH= NCH2R) and finally, a secondary amine. Similarly, the primary amine may react with the imine, forming an addition product which also is reduced to a secondary amine Finally, the secondary amine may react with either the aldehyde or the imine to give products which are reduced to tertiary amines.
Similar reactions may occur when the carbonyl compound employed is a ketone.
Question 1.
Ethanal on reaction with ammonia forms an imine
(X) which on reaction with nascent hydrogen gives
(Y) Identify ‘X’ and ‘Y’.
(A) X is CH3CH=NH and Y is CH3NH2
(B) X is CH3CHOHNH2 and Y is CH3CH2NH2
(C) X is CH3CHOHNH2and Y is CH3NH2
(D) X is CH3CH=NH and Y is CH3CH2NH2
Answer:
(D) X is CH3CH=NH and Y is CH3CH2NH2
Question 2.
Acetaldehyde is reacted with ammonia followed by reduction in presence of hydrogen as a catalyst. The primary amine so formed further reacts with ” acetaldehyde. The Schiff’s base formed during the reaction is:
(A) CH3CH=NHCH3
(B) CH3CH=NHCH2CH3
(C) CH3=NHCH2CH3
(D) CH3CH2CH=NHCH3
Answer:
(B) CH3CH=NHCH2CH3
Question 3.
The reaction of ammonia and its derivatives with aldehydes is called:
(A) Nucleophilic substitution reaction
(B) Electrophilic substitution reaction
(C) Nucleophilic addition reaction
(D) Electrophilic addition reaction
Answer:
(C) Nucleophilic addition reaction
Question 4.
![]()
The compound Q is:
(A) (CH3CH2CH2)3N
(B) (CH3CH2CH2)2N(CH2CH3)
(C) (CH3CH2)3N
(D) (CH3CH2)2NH
Answer:
(A) (CH3CH2CH2)3N
OR
Reductive alkylation of ammonia by means of an aldehyde in presence of hydrogen as reducing agents results in formation of:
(A) Primary amines
(B) Secondary amines
(C) Tertiary amines
(D) Mixture of all three amines
Answer:
(D) Mixture of all three amines






0 Comments